
Welcome!

Thank you for joining us today for the March 23rd, 2024, International Myeloma Foundation’s Regional Community Workshop –Seattle









Welcome!

Thank you for joining us today for the March 23rd, 2024, International Myeloma Foundation’s Regional Community Workshop –Seattle







March 23rd, 2024 - Agenda

9:00 – 9:15 AM
9:15 – 9:45 AM
Welcome & Introductions, Robin Tuohy
Myeloma 101, Andrew Cowan, MD
9:45 – 9:55 AM Q&A
9:55 – 10:40 AM
Taking the Reins of Your Multiple Myeloma Care, Donna Catamero, ANP-BC, OCN, CCRC
10:40 – 10:50 AM Q&A
10:50 – 11:00 AM
11:00 – 11:45 AM
Coffee Break
Frontline Therapy, Andrew Portuguese, MD
11:45 – 11:55 AM Q&A
11:55 AM – 12:40 PM LUNCH
12:40 – 1:00 PM

1:00 – 1:10 PM
1:10 – 1:30 PM
1:30 – 1:40 PM
1:40 – 2:25 PM
2:25 – 2:35 PM
2:35 – 2:45 PM
2:45 – 3:00 PM
March 2nd, 2024 – Agenda after lunch
Local Patient & Care Partner Panel, Sheri Baker, Patient & Jim Baker, Care Partner
Q&A
Maintenance Therapy, Andrew Portuguese, MD
Q&A
Relapsed Therapies & Clinical Trials, Andrew Cowan, MD
Q&A
Closing Remarks
Coffee / Network
The IMF provides FREE resources to help both patients and families.
Established in 1990, the IMF’s InfoLine assists over 4600 callers annually and answers questions across a wide variety of topics including:

Frequent topics:
Treatment questions along the spectrum of care
Clinical Trial access and understanding
Side effect management and health issues
Financial resources for myeloma-related expenses
Myeloma Specialist Referral contact information
Support group information
Caregiver Support






A core mission of the IMF is to provide thorough and cutting-edge education to the

















The IMF Support Group Team is Here For You!
• Support Groups Empower Patients & Care Partners with information, insight, & hope
• The IMF provides educational support to a network of over 150 myeloma specific groups

Support.myeloma.org



We are happy to help connect you with an existing support group or help form a new one! We assist with virtual, in-person, and hybrid options for meetings.
Reach out to us at
SGTeam@myeloma.org

Local Support Groups: You Are Not Alone!

Central Washington Blood Cancer Support Group
Meets in-person on the 1st Thursday of each month at 2pm Pacific Time
The Northwest Multiple Myeloma Fighters (Puget Sound)
Meets mostly virtually on the 4th Saturday of most months at 10AM Pacific Time
The Spokane, WA / Coeur d’Alene, ID Multiple Myeloma Education Network
Meets in-person on the 2nd
Providence Cancer Institute Multiple Myeloma Support Group (Portland, OR/ SW Washington)
Meets virtually on the 2nd
Wednesday of each month at 10AM Pacific Time


Tuesday of each month at 5:30PM Pacific Time
Treasure Valley Multiple Myeloma Support Group (Boise, ID)
Meets in a hybrid format on the 3rd Tuesday of each month at 6:30PM Mountain Time


Special interest groups are designed as a supplemental support for specific populations of patients, in addition to their local Support Groups
Las Voces de Mieloma
Designed for Spanish speaking patients only
Living Solo & Strong with Myeloma
Designed for patients without a care partner

Coming Soon!
Care Partners Only

Designed to address the needs of care partners only
Smolder Bolder
Created for people living with Smoldering Multiple Myeloma
MM Families

High Risk Multiple Myeloma
Designed to address the needs of the high-risk MM population
MGUS 4 Us
Created for people living with MGUS
For patients/care partners with young children


Fred Hutchinson Cancer Center
Seattle, WA
Andrew Cowan, MD• Review the basics of blood and cancer
• Define multiple myeloma and its key features
• Discuss the staging and classification of myeloma
• Outline the approach to therapy of myeloma
• Appreciate the importance of health disparities in myeloma
• The blood is an “organ” made up of both cells and liquid “plasma”
• Think of wine (red/white/rose)
1. Red Cells – carry Oxygen…trucks
2. White Cells – immune system…army
3. Platelets – help with clotting…ambulance
All produced in the blood factory = Bone Marrow

Multiple Myeloma* is a blood cancer that starts in plasma cells from the center of bones (bone marrow).
– This is where stem cells mature into red blood cells, white blood cells, and platelets
– Myeloma cells are abnormal plasma cells that make an abnormal antibody called “M protein”
– M = monoclonal (“identical” or cancerous)
* Myeloma is NOT a bone cancer or skin cancer (melanoma), it is a type of blood cancer.


• Cancer of plasma cells
• Healthy plasma cells produce immunoglobulins G, A, M, D, and E
• Myeloma cells produce abnormal immunoglobulin “paraprotein” or monoclonal protein (=M protein)

Bone marrow of patient with multiple myeloma
Image courtesy of American Society of Hematology
Kyle et al. Mayo Clin Proc. 2003;78:21-33;
1.8% of all cancers;
17% of hematologic malignancies in the United States
Most frequently diagnosed in ages
65 to 74 years (median, 69 years)
The average age of diagnosis of 4-5 years younger in African American and Hispanic patients
National MM Statistics
Approx 35,000 Estimated New Cases in 2023

Approx 13,000 Estimated Deaths in 2023
The Average Survival of patients with myeloma is IMPROVING!
Trends in MM Natural History by Race
MM Incidence
MM Mortality
Higher incidence in AA vs White patients:
• 15.9 vs 7.5 cases per 100,000 per year
Higher mortality in AA vs White patients:
• 5.6 vs 2.4 MM deaths per 100,000
5-year relative survival evolution from 1973 to 2005
MM Survival
• Survival for White patients increased significantly from 26.3% to 35%
• Survival for AA patients increased from 31% to 34.1%
The expected survival is nearly 10 years for all patients, but still less than 5 years in patients with high risk disease


• For example:
• IgG+kappa
• IgG+lambda
• IgA+kappa
• IgA+lambda
• etc…
• 80% of myeloma cases


Bence Jones protein
• 18% of all myeloma cases
• Renal failure more common in light chain multiple myeloma; creatinine >2 mg/dL in 1/3 of cases

protein present
• Less than 3% of cases of multiple myeloma
• Subtypes of MM are determined based on the kind of abnormal protein
IgG – 55%
IgA – 25%
IgD – 1-2%
IgM – 1%
Light Chain Disease only – 20%
Non Secretors 1-2 %

 M spike in gamma region
M spike in gamma region
Condition
Premalignant Malignant
MGUS1-4 (Monoclonal Gammopathy of Undetermined Significance)
SMM1-5,8 (Smoldering Multiple Myeloma) Active Multiple Myeloma6-8
* In clinical trial (preferred) or offer treatment for those likely to progress within 2 years
1. Kyle RA, et al. N Engl J Med. 2007;356:2582-90.
2. International Myeloma Working Group. Br J Haematol. 2003;121:749-57.
3. Jagannath S, et al. Clin Lymphoma Myeloma Leuk. 2010;10(1):28-43.
4. Kyle RA, et al. Curr Hematol Malig Rep. 2010;5(2):6269.
5. Mateos M-V, et al. Blood. 2009;114:Abstract 614.
6. Durie BG, Salmon SE. Cancer. 1975;36:842-854.
7. Durie BG, et al. Leukemia. 2006;20(9):1467-1473.
8. Rajkumar SV, et al. Lancet Oncology 2014; 15:e538e548.





 Kyle RA. Mayo Clin Proc. 2003;78:21-33.
Fatigue Anemia
Bone Pain
Kyle RA. Mayo Clin Proc. 2003;78:21-33.
Fatigue Anemia
Bone Pain
Clonal bone marrow ≥10% or bony/extramedullary plasmacytoma
AND any one or more Myeloma-Defining Events
Now SLiM CRAB
C
alcium elevation
R A B
enal complications
Clonal bone marrow ≥60% BM
BM, bone
nemia one disease
FLC MRI
sFLC ratio >100
>1 focal lesion by MRI
• S (60% Plasmacytosis)
• Li (Light chains I/U >100)
• M (MRI 1 or more focal lesion)
• C (calcium elevation)
• R (renal insufficiency)
• A (anemia)
• B (bone disease)

Rajkumar et al. Lancet Oncol. 2014;15:e538-e548. Kyle et al. Leukemia 2010;24:1121-1127.
marrow; FLC, free light chain; MRI, magnetic resonance imaging; sFLC, serum free light chain.



Low Blood Counts
• May lead to anemia and infection
• Anemia is present in 60% at diagnosis
Decreased Kidney Function
• Occurs in over half of myeloma patients
Weakness
Fatigue Infection
Weakness
Bone Damage
• Affects 85% of patients
• Leads to fractures
Bone Turnover
• Leads to high levels of calcium in blood (hypercalcemia)
Bone pain
Loss of Appetite & Weight loss
About 10% to 20% of patients with newly diagnosed myeloma will not have any symptoms.

CBC
Counts the number of red blood cells, white blood cells, and platelets

CoMP
Measures levels of albumin, calcium, and creatinine to assess kidney and liver functions, bone status ,and the extent of disease

Beta2 MicroG
Determines the level of a protein linked to MM and kidney function: USED FOR STAGE

LDH Lactate Dehydrogenase
Determines the level of myeloma cell production and extent of MM : USED FOR STAGE

Serum Protein EP Detects the presence & level of M protein = how much myeloma. No Heavy Chain = No M-Spike

Immuno Fixation
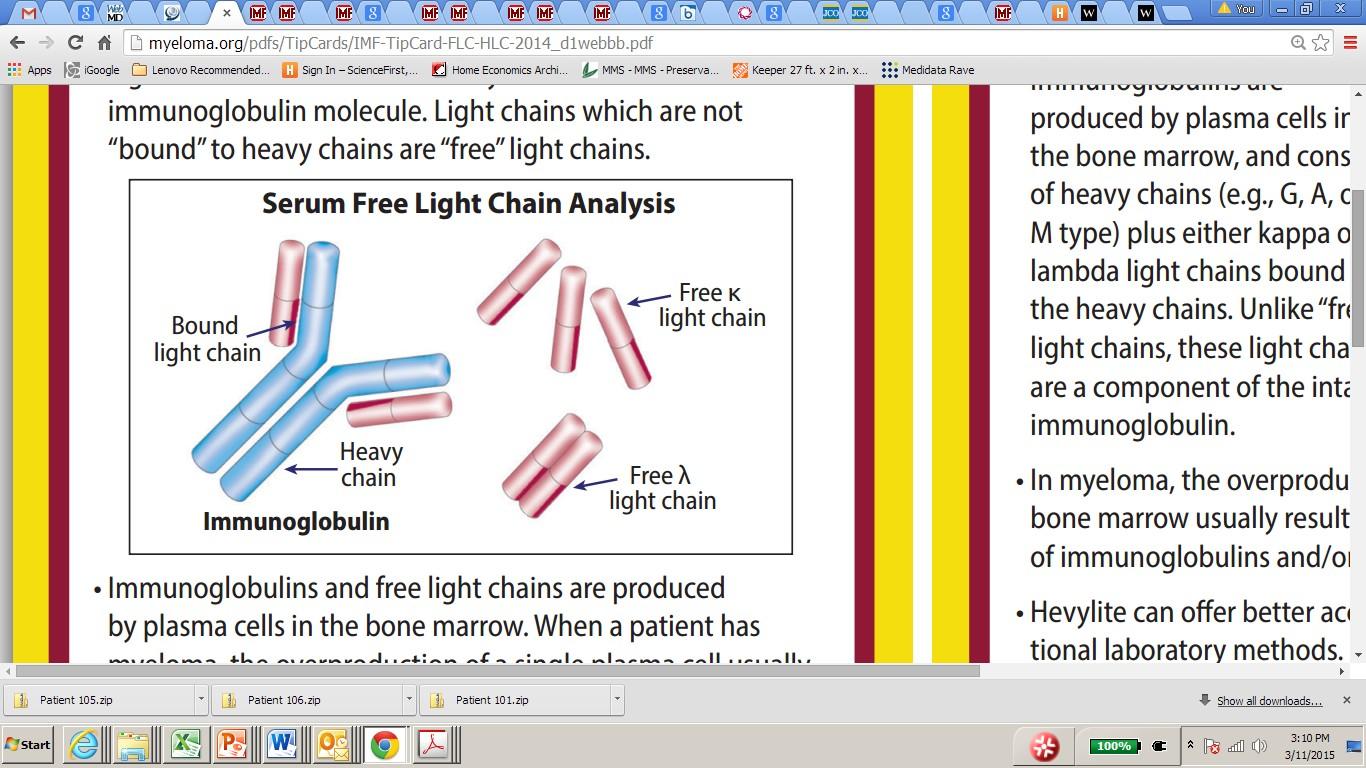
Serum Free Light Chain
Identifies the type of abnormal antibody proteins: IgG, IgA, IgM

Measures myeloma free light chains (kappa or lambda) in blood = how much myeloma

Urine Protein EP Detects Bence-Jones proteins (otherwise known as myeloma light chains) in urine (to determine if it’s present or not present)

24-hr Urine Analysis
24 hours of urine collected to test the presence and levels of Bence Jones protein in the urine = how much myeloma
Test Name
CBC + differential
Complete metabolic panel
Beta-2 Microglobulin (B2M)
Lactate Dehydrogenase (LDH)
Serum Immunofixation and Protein
electrophoresis (SPEP+IFE)
Immunoglobulins (G, A, M, D, E)
Free light chain assay with kappa/lambda ratio
Urine immunofixation & protein
electrophoresis (UPEP+IFE)
What it means

Hemoglobin, WBC, Platelets
Creatinine, Calcium, Albumin, Liver function
Part of staging and risk stratification
Measures the level of normal and clonal protein
Identifies the type of clonal protein

Measures the level of normal and clonal protein
Identifies the type of clonal protein

Oncol. 2014;15:e538-3548. Ghobrial IM, et al. Blood. 2014;124:3380-3388; mSMART.org; NCCN.org This




Imaging:
– Skeletal survey: Series of X-rays; less sensitive than other techniques
– Whole body low dose (CTWB-LD CT )
– Positron Emission Tomography (PET/CT)
– Magnetic Resonance Imaging (MRI)










Bone marrow biopsy & aspirate
• Bone marrow plasma cells (%)
• Congo Red staining if concern for AL-Amyloid
Bone marrow genetics
• Cytogenetics
• Fluorescence in situ hybridization (FISH)
• Next generation sequencing (NGS)





High Risk FISH Results
Deletions Translocations Gain
1p17pp53 mutation t(4;14) t(14;16) t(14;20) 1q+


Fluorescence in situ hybridization (FISH) and Chromosomal abnormalities

Image Credit: IMF Patient Handbook



Myeloma Stage:
Staging refers to the degree to which the cancer has progressed. Most important at time of diagnosis.
Stage 1
β2-microglobulin under 3.6 mg/L
Normal
Lactate Dehydrogenase (LDH)
Stage 2
β2-microglobulin
Between 3.5 & 5.4mg/L
Stage 3
β2-microglobulin over 5.5 mg/L
NO High Risk Cytogenetics (FISH) AND
NO High Risk Cytogenetics (FISH)
HIGH Lactate Dehydrogenase (LDH)
High Risk Cytogenetics (FISH)
Deletion 17thchromosome
Translocation 4th and 14th
Translocation 14th and 16th
Translocation 14th and 20th AND/OR
Treatment Planning is the process of thinking about the treatment steps you can take with your doctor, based on your goals and preferences.
Treatment decisions are based on:
• The results of biomarker tests, cytogenetic (FISH) test, and the stage of multiple myeloma
• Your values, goals, and preferences
• Your age
• Your health and symptoms (if you have kidney disease, heart disease, anemia, or other issues)
• Your medical history and past treatments for multiple myeloma
Transplan
t Eligible Patients
Transplan t
Ineligible Patients
Initial Therap y
Transplant (ASCT) Maintenan
ce Treatme nt of Relapse d disease
Consolidation / Maintenance
Continued therapy
Everyone
Supportive Care
HCP
Clinical Experience


Your Preference TREATMENT DECISION
Research Results
Philippe Moreau. ASH 2015.




• You have the right to get a second opinion. Insurance providers may require second opinions.
• A second opinion can help you:
– Confirm your diagnosis
– Give you more information about options
– Talk to other experts
– Introduce you to clinical trials

– Help you learn which health care team you’d like to work with, and which facility


ASYMPTOMATIC
MGUS or SMOLDERING MYELOMA ACTIVE MYELOMA
SYMPTOMATIC REFRACTORY RELAPSE
RELAPSE

Clone 1.1
Clone 1.2
Clone 2.1
Clone 2.2
Misc
PLATEAU REMISSION
Therapy
MGUS = monoclonal gammopathy of undetermined significance
Adapted from Dr. Brian Durie and Keats JJ, et al. Blood. 2012;120:1067-1076.
Time
Class Drug Name Abbreviation Administration

IMiD
immunomodulatory drug
Proteasome inhibitor
Chemotherapy
Steroids
Monoclonal Antibodies
Pomalyst (pomalidomide) P or Pom
Oral Revlimid (lenalidomide) R or Rev
Thalomid (thalidomide) T or Thal
Velcade (bortezomib)
Kyprolis (carfilzomib) C or K or Car
V or Vel or B Intravenous (IV) or subcutaneous injectionSC (under the skin)
Ninlaro (ixazomib) N or I
Cytoxan (cyclophosphamide) C
Alkeran or Evomela (melphalan) M or Mel
Decadron (dexamethasone) Dex or D or d
Prednisone P or Pred
Darzalex (daratumumab)
Sarclisa (isatuximab)
Empliciti (elotuzumab)
Oral
Oral or intravenous
Oral or intravenous
Dara Isa Elo Intravenous (IV or SC)
XPO1 Inhibitors Xpovio (selinexor) X or Sel Oral

Class
Drug Name
Abbreviation Administration
Peptide Drug Conjugate* Pepaxto (Melphalan Flufenamide) Melflufen Intravenous
BCMA Targeted Antibody Drug Conjugate (ADC)* Blenrep (belantamab mafodotin) Bela, Belamaf, or B Intravenous
Abecma (idecabtagene vicleucel)
CAR T Cell therapy
Bispecific Antibodies
Carvycti (ciltacabtagene vicleucelel) Cilta-cel
Tecvayli (teclistimab)
Talvey (Talquetamab)
Elrexfio (Elranatamab)
Ide-cel Intravenous (IV) or subcutaneous injectionSC (under the skin)
Tec Talq Elra SC or IV ??? MORE
* these agents are currently off the market but available through special programs
Antibody-dependent Cellular cytotoxicity (ADCC)
ADCC Effector cells: MM FcR
• Lucatumumab or Dacetuzumab (CD40)
• Elotuzumab (CS1; SLAMF7)
• Daratumumab, SAR650984/Isatuximab (CD38)
• XmAb5592 (HM1.24)
Complement-dependent Cytotoxicity (CDC)
CDC MM C1q C1q
Apoptosis/growth arrest via targeting signaling pathways
• Daratumumab
• SAR650984/Isatuximab (CD38)
• huN901-DM1 (CD56)
• nBT062-maytansinoid (CD138)
• Siltuximab (1339) (IL-6)
• BHQ880 (DKK1)
• RAP-011 (activin A)
• Daratumumab, SAR650984/Isatuximab (CD38)

Effector cell

• Incorporates 2 antibody fragments to target and bind both tumor cells and T cells
• Brings target-expressing MM cells and T cells into close proximity, enabling T cells to induce tumor-cell death

Antibody-Drug
Bispecific Molecule Targets Vary
Talquetamab GPRC5d CD3
Cevostamab FcRH5 CD3
“Off the Shelf” Advantage
• No manufacturing process, unlike CAR T-cell therapy (but like ADC/belantamab therapy)
• Thus, no delay between decision to treat and administration of drug
Image Source: Shah N, et al. Leukemia. 2020;34:985–1005. Creative Commons License: CC BY 4.0.
Bi-Specific Antibodies
Talquetamab
CAR-T


GPRC5D

SLAMF7

FcRH5



Antibody Drug
Elotuzumab
Bi-Specific Antibodies

Bi-Specific Antibodies
CAR-T


BCMA
CD38
Antibody Drug
Daratumumab and Darzalex Faspro
Isatuximab
TAK-079
MOR202


Immune Therapies
Ide-cel CAR-T
Cilta-cel CAR-T
Teclistamab
Other Bi-Specific Antibodies
Other CAR-Ts
Bortezomib
Lenalidomide
Carfilzomib
Panobinostat
Daratumumab Ixazomib
Now
VTD VRD KRD D-VMP DRD D-VRD SCT +/- More induction
Front line treatment
New
Induction Consolidation
Isa-VRD D-KRD Isa-KRD
CAR T or Bispecifics?
Lenalidomide
Bortezomib Ixazomib
Lenalidomide + PI Carfilzomib
Combinations
Maintenance
Post consolidation
Pomalidomide Selinexor
Elotuzumab
Isatuximab
Idecabtagene autoleucel
Ciltacabtagene autoleucel
Teclistamab
Talquetamab
Elranatamab
Rescue Relapsed
Daratumumab?
Novel CAR T Cell Therapies
Bispecific/Trispecific Antibodies
CelMod Agents
Venetoclax?
Modakafusp
Multiple small molecules
1.There is a longer time from symptoms to diagnosis among African Americans
2.African Americans are younger by about 5 years on average at diagnosis
3.MM and MGUS are more than 2x as common in African Americans
4.African Americans are less likely to receive the four T’s: Transplant, Triplets, Trials and CAR T
5.African Americans have biologic differences with more t(11;14) and less high-risk cytogenetics with deletion 17p
6.Survival outcomes in African Americans are HALF of what is seen in White Americans
7.African Americans can achieve equal or better outcomes when they receive therapy

The core vision of this initiative is to improve the short- and long-term outcomes of African American patients with myeloma. We want to empower patients and communities to change the course of myeloma…
Engage the community to increase awareness and provide support
Enhance access to optimal care by educating myeloma providers about the disparity and how to reduce it

Shorten the time to diagnosis by educating primary care providers to recognize the disease and order the right tests
Community events

Educational Postcards
Publications Mentorship of Medical Students














Donna Catamero, ANP- BC, OCN, CCRC
Ichan School of Medicine, Mount Sinai
IMF Nurse Leadership Board Member


STABLE OF TREATMENT
Myeloma and treatment options, side effects, symptom management, & supportive care

FINDING YOUR GAIT
Know your care team & be an empowered patient

GOING THE DISTANCE
Healthful and meaningful living
Treatment options, side effects, symptom management, and supportive care





Rapid and effective disease control
Durable disease control
Improved overall survival
Minimize side effects
Promote good quality of life
Prevent disease- and treatment-related side effects
Optimize symptom management
Promote quality of life
Discuss your goals and priorities with your healthcare team.

FRONTLINE
MAINTENANCE
Velcade® (bortezomib)
Darzalex® (daratumumab)
Velcade® (bortezomib)
Ninlaro® (ixazomib)
Kyprolis® (carfilzomib)
RELAPSE
PENDING FDA
APPROVAL
Ninlaro® (ixazomib)
Darzalex® (daratumumab) in clinical trial
Darzalex® (daratumumab)
Empliciti® (elotuzumab)
Sarclisa® (Isatuximab)
Revlimid® (lenalidomide)
Thalomid® (thalidomide)
Revlimid® (lenalidomide)
Thalomid® (thalidomide)
Revlimid® (lenalidomide)
Pomalyst® (pomalidomide)
• CelMods ‒ Iberdomide
‒ Mezigdomide
Dexamethasone
Prednisone
Prednisolone
SoluMedrol Melphalan Cyclophosphamide
Dexamethasone
Prednisone
Prednisolone
SoluMedrol Melphalan Cyclophosphamide
Bendamustine
Elrexfio™ (elranatamab)
Tecvayli® (teclistamab)
Talvey™ (talquetamab)
Other Bispecific Antibodies
‒ Cevostamab


Others
Melphalan + ASCT
Melphalan + ASCT
CAR-T
− Ide-Cel
− Cilta-Cel Xpovio® (Selinexor)
Doxil (liposomal doxorubicin)
Other CAR-T Venclexta® (venetoclax):BCL2 inhibitor for t(11;14) Blenrep (belantamab mafodotin)*: antibody drug conjugate
NOTED SIDE
EFFECTS Neuropathy
Carfilzomib: Cardiac Infusion reaction
DVT/PE See steroid slide Myelosuppression
CRS and neurotoxicity; infection risk
Talvey: skin/nail/GI Infection risk
CAR-T: CRS and neurotoxicity
Myelosuppression, GI Xpovio: low sodium Blenrep: eye-related

Measuring treatment response
Determining Transplant Eligibility
Insurance authorization
Collecting stem cells
Duration:
Approximately 2 weeks
Location:
Transplant Center



High Dose Chemotherapy, stem cell infusion Supportive Care Engraftment
POSTTRANSPLANT
Restrengthening
Appetite recovery
“Day 100” assessment
Begin maintenance therapy
Duration: Approximately 3-4 weeks Location: Transplant Center

Duration: Approximately 10-12 weeks
Location: HOME
Upfront stem cell transplant remains the standard of care for eligible patients

Ask for a referral to CAR Tcell center as soon as it is possible as next treatment option (ie, before relapse)



No driving for 8 weeks
“One & Done” with continued monitoring
T-Cell Collection
Manufacturing takes ≈ 4 to 6 weeks
Bridging therapy may be needed
• Away from home
• Often some hospital stay
• Care Partner needed
• Side effect management
• CRS, ICANS
• Low blood counts
• Fatigue and fever
• Some patients need ongoing transfusion support
Horse of Another Breed:

•Different bispecific antibodies have differences in efficacy, side effects
– Available after 4 prior lines of therapy (or clinical trial)
– About 7 in 10 patients respond
– Off-the-shelf treatment; no waiting for engineering cells
– CRS and neurotoxicity
– Risk of infection
•BCMA target: greater potential for infection
– Tecvayli® (teclistamab)
– Elrexfio™ (elranatamab)
•GPRC5D target: potential for skin and nail side effects, GI issues of taste change, anorexia and weight loss
– Talvey™ (talquetamab)
Bispecific Antibodies MM cell death Target CD3

BISPECIFIC ANTIBODIES
Cytotoxic cytokines






CAR T and Bispecific Antibodies: Unique Side Effects
Fever
Confusion
Weakness
Fatigue


Headache
CRS is a common but often a mild & manageable side effect
Diarrhea
Nausea / vomiting
Shortness of Breath
CAR = chimeric antigen receptor; CRS = cytokine release syndrome.
Oluwole OO, Davila ML. J Leukoc Biol. 2016;100:1265-1272. June CH, et al. Science. 2018;359:1361-1365. Brudno JN, Kochenderfer JN. Blood. 2016;127(26):3321-3330.
Brudno JN, Kochenderfer JN. Blood Rev. 2019:34:45-55. Shimabukuro-Vornhagen, et al. J Immunother Cancer. 2018;6:56. Lee DW, et al. Biol Blood Marrow Transplant. 2019;25:625-638.


Neurotoxicity
Tremors


Steroids enhance the effectiveness of other myeloma therapies
Do not stop or alter your dose of steroids without discussing it with your provider


• Irritability, mood swings, depression
• Difficulty sleeping (insomnia), fatigue
•Consistent schedule (AM vs. PM)
•Take with food
•Stomach discomfort: Over-the-counter or prescription medications
•Medications to prevent shingles, thrush, or other infections
• Blurred vision, cataracts
• Flushing/sweating
• Increased risk of infections, heart disease
• Stomach bloating, hiccups, heartburn, ulcers, or gas
• Muscle weakness, cramping
• Weight gain, hair thinning/loss, skin rashes
• Increased blood pressure, water retention
• Increased blood sugar levels, diabetes
[P]reventing
Infection remains the leading cause of death in patients with multiple myeloma. Several factors account for this infection risk, including the overall state of immunosuppression from multiple myeloma, treatment, age, and comorbidities (e.g., renal failure and frailty).
IMWG Consensus guidelines and recommendations for infection prevention in multiple myeloma; Lancet Haematol.2022;9(2):143–161.
Infection Prevention Tips
Good personal hygiene (skin, oral)
Environmental control (avoid crowds and sick people; use a high-quality mask when close contact is unavoidable)
IMWG = International Myeloma Working Group; HCP = healthcare provider.
RAJE NS, et al. Lancet Haematol.2022;9(2):143–161. IMF Nurse Leadership Board ONS Symposia 2023.




Report fever of more than 100.4°F, shaking chills even without fever, dizziness, shortness of breath, low blood pressure to HCP as directed.
As recommended by your healthcare team:
Immunizations: Flu, COVID, RSV & and pneumococcal vaccinations; avoid live vaccines
Preventative and/or supportive medications (next slide)
Medications Can Reduce Infection Risk
Type of Infection Risk
Herpes virus reactivation (HSV/VZV); CMV reactivation
Bacteremia, pneumonia, and urinary tract infection
PJP (P jirovecii pneumonia)
Fungal infections (aspergillus)
IgG < 400 mg/dL or recurrent infections
ANC < 500 cells/μL
ANC
COVID-19


Medication Recommendation(s) for Healthcare Team Consideration
Acyclovir prophylaxis
Consider prophylaxis with levofloxacin
Consider prophylaxis with trimethoprim-sulfamethoxazole
Consider prophylaxis with fluconazole
IVIg
GCSF 2 or 3 times/wk (or as frequently as needed) to maintain ANC > 1000 cells/μL and maintain treatment dose intensity
Antiviral therapy if exposed or positive for covid per institution recommendations
Some people receiving BCMA-targeting therapies have experienced infections that are less common like CMV, PJP and fungal infections
RAJE NS, et al. Lancet Haematol.2022;9(2):143–161.






Talvey™ (talquetamab): Common Generally Mild and Painless Skin and Nail Side Effects


Body Rash:
•Prevent dry skin; apply lotion
– Ammonium lactate 12% lotion
•Steroids:
– Topical for grades 1-2,
– Systemic and topical for Grade 3 and dose hold
•Antihistamines, as needed

.
Nail Changes:
•Keep your nails short and clean. Watch for “catching and tearing”
•Apply a heavy moisturizer like Vaseline or salve. Wear cotton hand coverings to bed
•A nail hardener may help with thinning
Photos: Mount Sinai Hospital, NY, NY IMF Nurse

•Tell the team if you have signs of a fungal infection, like thickened or discolored nails

Dexamethasone oral solutions “swish and spit” have been tried but with no proven benefit yet. Sour citrus or candies before meals are also recommended.
OTC dry mouth rinse, gel, spray are recommended. Advise patients to avoid hot beverages.
Glossitis

EARLY initiation of nystatin or Mycelex is key to manage symptoms.
• Weight loss and anorexia are associated with taste changes. Nutritionist involvement and dietary modifications are recommended to support patients. Appetite stimulant with Marinol, if indicated, can also be utilized.
Dietary modifications with small bites, eating upright, and sips with food can help manage symptoms.
• Education and emotional support are key strategies to manage oral toxicities.
Prevention & Management
Diarrhea may be caused by medications and supplements
– Laxatives, antacids with magnesium
– Antibiotics, antidepressants, other (check with provider, pharmacist)
– Supplements: milk thistle, aloe, cayenne, saw palmetto, ginseng
Avoid caffeinated, carbonated, or heavily sugared
beverages
Take anti-diarrheal medication if recommended


Constipation may be caused by medications and supplements
– Opioid pain relievers, antidepressants, heart or blood pressure medications (check with provider, pharmacist)
– Supplements: Calcium, Iron, vitamin D (rarely), vitamin B-12 deficiency
Increase fiber
•Fruits, vegetables, high fiber whole grain foods
•Fiber binding agents – Metamucil®, Citrucel®, Benefiber®



Pain Prevention and Management
Sources of pain include bone disease, neuropathy and medical procedures
•Management
– Prevent pain when possible
• Bone strengtheners to decrease fracture risk
• Antiviral to prevent shingles
• Sedation before procedures
– Interventions depend on source of pain



Tell your healthcare provider about any new bone or chronic pain that is not adequately controlled
• May include medications, activity, surgical intervention, radiation therapy, etc
• Complementary therapies (Mind-body, medication, yoga, supplements, acupuncture, etc)
• Scrambler therapy for neuropathy
Sufficient Sleep: Important for Good Health
•Adequate rest and sleep are essential to a healthful lifestyle
•Shortened and disturbed sleep cause
– Increased heart-related death
– Increased anxiety
– Weakened immune system
– Worsened pain
– Increased falls and personal injury
•Things that can interfere with sleep
– Medications: steroids, stimulants, herbal supplements
– Psychologic: fear, anxiety, stress
– Physiologic: sleep apnea, heart issues, pain

• Sleep hygiene is necessary for quality nighttime sleep and daytime alertness
– Engage in exercise but not too near bedtime
– Increase daytime natural light exposure
– Avoid daytime napping
– Establish a bedtime routine - warm bath, cup of warm milk or tea
•Associate your bed ONLY with sleep
– Avoid before bedtime:
• Caffeine, nicotine, alcohol and sugar
• Large meals and especially spicy, greasy foods
• Computer screen time
•Sleep aid may be needed
Rod NH et al 2014. PloS one. 9(4):e91965; Coleman et al. 2011. Cancer Nurs. 34(3):219-227.National Sleep Foundation. At: http://sleepfoundation.org/ask-the-expert/sleep-hygiene Mustian et al. Journal of clinical Oncology. Sep 10 2013;31(26):3233-3241; Stan DL, et al. Clin J Oncol Nurs. Apr 2012;16(2):131-141; Zeng Y et al., Complementary therapies in medicine. Feb 2014;22(1):173-186.
Peripheral Neuropathy Management


Peripheral neuropathy happens when there is damage to nerves in the extremities (hands, feet, limbs). Damage can be the result of myeloma, treatment or unrelated conditions (i.e. diabetes).
Symptoms:
•Numbness
•Tingling
•Prickling sensations
•Sensitivity to touch
•Burning and/or cold sensation
•Muscle weakness
Prevention / management:
•Bortezomib once-weekly or subcutaneous administration
•Massage area with cocoa butter regularly
•Neuroprotective Supplements:
– B-complex vitamins (B1, B6, B12)
– Green tea
•Safe environment: rugs, furnishings, shoes
Report symptoms of peripheral neuropathy early to your health care provider; nerve damage from neuropathy can be permanent if unaddressed
If neuropathy worsens, your provider may:
•Adjust your treatment plan
•Prescribe oral or topical pain medication
•Suggest physical therapy
•Risk Factors
– Active multiple myeloma (light chains, high calcium)
– Other medical issues (ex: Diabetes, dehydration, infection)
– Medications (MM treatment, antibiotics, contrast dye)
•Prevention
– Stay hydrated – drink water
– Avoid certain medications when possible (eg, NSAIDs), dose adjust as needed
•Treatment
– Treatment for myeloma
– Hydration
– Dialysis




Many myeloma patients will experience kidney issues at some point; protecting your kidney function early and over time is important

98.8%


Fatigue is the most commonly reported symptom.
Sources include anemia, pain, reduced activity, insomnia, treatment toxicity, bone marrow suppression
>35% of patients
≈25% of patients
Often, people do not share these symptoms with their provider. Talk to your provider about symptoms that are not well controlled or if you have thoughts of self harm. Help is available.
Supportive Medications
DVT/PE Prevention
Blood thinners
Ex: Aspirin, DOACs


Bone Health Fatigue Anxiety
Bone Strengthening Agents
Calcium
Vitamin D
Stimulant medications
Anti-depressants
Anti-anxiety
Non-medication
Therapies
Compression stockings
Radiation Surgery
Immobilization
Physical therapy
Relaxation
Meditation
Therapy
Relaxation
Meditation
Activity
Lifestyle Options
Stop smoking
Weight loss
Activity
Activity
Improved sleep Activity
Improved sleep
Faiman B, et al. CJON. 2017;21(5)suppl:19-36. Dimopoulous M, et al. Leukemia. 2009;23(9):1545-56. Brigle K, et al. CJON. 2017;21(5)suppl:60-76. Faiman B, et al. CJON. 2017;21(5)suppl:19-36. Faiman B, et al. CJON. 2011;15suppl:66-76. Miceli TS, et al. CJON. 2011;15(4)suppl:9-23.
Financial Burden


Financial burden comes from
•Medical costs
– Premiums
– Co-payments
– Travel expenses
– Medical supplies
•Prescription costs
•Loss of income
– Time off work or loss of employment
– Caregiver time off work

•Funding and assistance may be available
– Federal programs, IRA & Medicare “Extra Help”
– Pharmaceutical support
– Non-profit organizations
– Websites:
• Medicare.gov
• SSA.gov
• LLS.org
• Rxassist.org
• NeedyMeds.com
• HealthWellFoundation.org
• Company-specific website

Be an empowered patient; engage in your care

YOU are central to the care team
Be empowered
Ask questions, learn more
Express your goals/values/preferences






Ask for time to consider options
Communicate with your team
Understand the roles of each team member and who to contact for your needs
Arrive at a treatment decision together
Create a support network










You






Don’t Get Left in the Dust:
Communicate How You Feel With Your Team
Unmanaged Myeloma can cause:
Calcium elevation
Renal dysfunction
Low blood counts
Infection Risk
Blood clots
Bone pain
Neuropathy
Fatigue


Your team may be able to help, but only if they know how you feel.

Side Effects of Treatment can cause:
GI symptoms
Renal dysfunction
Low blood counts
Infection Risk
Blood clots
Neuropathy
Fatigue































If you want to go fast, go alone, if you want to go far, go
•Care partners may help in many ways including medical appointments, managing medication, daily living, physical assistance, emotional support, myeloma knowledge, healthy lifestyle, patient advocacy, financial decisions
•Care partners can be a spouse, close relative, a network of people (family, friends, neighbors, church members, etc)


•Caring for the Care Partner
together
African Proverb
– Recognize that caregiving is difficult/stressful
– Encourage care partners to maintain their health, interests, and friendships

– The IMF has information and resources to help care partners


Form A Posse: Build Strong Social Ties & Cultivate a Sense of Belonging
•Multiple studies demonstrate that strong social ties are associated with
– Increased longevity including people with cancer
– Improved adherence to medical treatment leading to improved health outcomes
– Lower risk of developing cardiovascular diseases
– Increased sense of purpose and life satisfaction
– Reduced stress and anxiety
– Improved mood and happiness
– Enhanced resilience
Martino J, et al. Am J of Lifestyle Med. 2015;11(6):466-475.
Yang YC, et al. Proc Natl Acad Sci U S A. 2016;113(3):578-583.
Pinquart M and Duberstein PR. Crit Rev Oncol Hematol. 2010; 75(2):122–137.


•Strategies for enhancing social connection
– Deepen existing relationships with family, friends, and loved ones
– Build new relationships by participating in a support group, joining clubs or organizations, or volunteering
Tip: Start with small steps outside your comfort zone. Call a loved one you haven’t spoken to in a while. Invite a person you’d like to know better for lunch, coffee, or a walk.

Hetherington C. Healthnews.

https://healthnews.com/longevity/healthspan/social-connection-andlongevity/#:~:text=Research%20consistently%20demonstrates %20that%20people,of%20fulfillment%20in%20your%20life. Accessed Feb 1 2024.
Maintain Good Health
Have a Primary Care Doctor
Have Recommended Health Screenings
• Blood pressure
• Cholesterol
• Cardiovascular disease
• Diabetes
• Colonoscopy
• Vision
• Hearing
• Dental checkups & cleaning
• Women specific: mammography, pap smear
• Men specific: prostate
Maintain a healthy weight
Good nutrition
Activity or exercise
Sufficient sleep
An ounce of prevention is worth a pound of cure.

 Benjamin Franklin
Benjamin Franklin
Faiman B, et al. CJON. 2017;21(5)suppl:19-36. Dimopoulous M, et al. Leukemia. 2009;23(9):1545-56.

Brigle K, et al. CJON. 2017;21(5)suppl:60-76. Faiman B, et al. CJON. 2017;21(5)suppl:19-36. Faiman B, et al. CJON. 2011;15suppl:66-76. Miceli TS, et al. CJON. 2011;15(4)suppl:9-23.










Andrew Portuguese, MD
Fred Hutchinson Cancer Center
Seattle, WA
• Review the importance of DEPTH of response in early treatment of myeloma and the increasing use of MRD testing
• Discuss emerging approaches in transplant eligible patients, including quadruplet therapy and stem cell transplantation
• Outline the approach to a patient not going to transplant and how to optimize continuous therapy
Treatment
>1 Billion
>1 Trillion D i s e a s e B u r d e n ( # o f m y e l o m a c e l l s )
>10 Million 1 myeloma cell in 100K to 1 million normal cells


Symptomatic Myeloma
At diagnosis
Partial response

50% reduction in M protein

Very good partial response
90% reduction in M protein immunofixation positive only

Complete remission No M-protein immunofixation negative

Minimal Residual Dis Flow Cytometry

Minimal Residual Dis
Next Generation Molecular testing
MRD refers to the persistence of residual tumor cells after treatment and is responsible for relapse1
Current techniques can detect MRD with a sensitivity of 10-6 for MM cells2


Newly Diagnosed MM and Risk Stratified
Factors to be considered for ASCT
Age, performance status (PS), comorbidities (R-MCI score, HCT-Cl) and organ function
ASCT Eligible
ASCT Ineligible
1. Most patients will be given a combination of drugs to control the disease quickly
2. We don’t “save the best for last” because early therapies have a long term effect on survival
3. We seek a DEEP and DURABLE response
4. We mix and match from the 3 major classes of drugs and add steroids:
Proteasome Inhibitors – most often botezomib (Velcade)
Immunomodulatory Drugs – lenalidomide (Revlimid)
Monoclonal Antibodies – daratumumab (Darzalex)
5. We decide early on whether or not someone will have a stem cell transplant
Induction
-Patients aged 18-65 yrs with symptomatic newly diagnosed MM following 1 cycle of RVD -56 sites within the United States from 2010 to 2018
RVD cycles 2-3 (n = 357)
RVD cycles 2-3 (n = 365)
End Points of Study and Follow-up
Consolidation
RVD cycles 4-8
ASCT: Melphalan 200 mg/m2
+ Stem Cell Support (n = 310)
RVD cycles 4-5
Maintenance Until Progression
R (n = 291)
R (n = 289)
• Primary end point: progression-free survival (time to next relapse)
• Secondary end points included:
• Response rates, overall survival, quality of life, and adverse events
• Follow-up on participant status : median of 6 years

Paul
HR 1.53 (1.23–1.91), p<0.0001
Data cut off:12/12/21

Median follow-up 76 months
*p-value adjusted using Bonferroni’s correction to control overall family-wise error rate for secondary outcomes

• ASCT remains very relevant and important in prolonging PFS in younger and eligible patients
• BUT it may not be mandatory in all eligible patients upfront
• As with other agents, we INDIVIDUALIZE the sequencing patterns
• ASCT does carry genuine toxicity, short term and long term
• We may become callous to these toxicities
• Maintenance therapy remains an important part of myeloma therapy
Induction Consolidation Maintenance
VRd
V: 1.3 mg/m2 SC
Key eligibility criteria
• Transplanteligible NDMM
• Age 18-70 years
• ECOG PS ≤2 1 : 1 r a n d o m i z a t i o n ( N = 7 0 9 ) a
Days 1, 4, 8, 11
R: 25 mg PO Days 1-21
d: 40 mg PO/IV
Days 1-4, 9-12
D-VRd
DARA: 1,800 mg
SCb
QW Cycles 1-2
Q2W Cycles 3-4
VRd administered as in the VRd group
4 cycles of 28 days
Primary endpoint: PFSc
T r a n s p l a n t
VRd V: 1.3 mg/m2 SC
Days 1, 4, 8, 11
R: 25 mg PO Days 1-21
d: 40 mg PO/IV
Days 1-4, 9-12
D-VRd
DARA: 1,800 mg
SCb
Q2W
VRd administered as in the VRd group
2 cycles of 28 days
Key secondary endpoints: Overall ≥CR rate,c overall MRD-negativity rate,d OS
D-R

R: 10 mg PO Days 1-28 until PD
DARA: 1,800 mg
SCb Q4W
R: 10 mg PO
Days 1-28
MRD positiv e MRD negati ve R
Continue D-R until PD
Discontinue DARA therapy only
28-day cycles
Discontinue DARA therapy only after ≥24 months of D-R maintenance for patients with ≥CR and 12 months of sustained MRD negativity
Abstracts Session, ASH 2023. Accessed from: https://ash.confex.com/ash/2023/webprogram/Paper191911.html
Restart DARA therapy upon confirmed loss of CR without PD or recurrence of MRD



Late-breaking Abstracts Session, ASH 2023. Accessed from: https://ash.confex.com/ash/2023/webprogram/Paper191911.html


Late-breaking Abstracts Session, ASH 2023. Accessed from: https://ash.confex.com/ash/2023/webprogram/Paper191911.html



NDMM N=662

Key eligibility criteria1
Age 18–70 years
NDMM and eligible for HDT and ASCT
Induction phase (3 x 6-week cycles)
Isa
Isa (IV) 10 mg/kg Cycle 1
Cycle 2–3
Bor (SC) 1.3 mg/m²
Len (PO) 25 mg
Dex (PO) 20 mg
GMMG
Maintenance phase (4-week cycles)
Isa + R R
3 years or PD
Isa (IV) 10 mg/kg: Cycle
(PO) 10 mg increased to 15 mg after 3 months
Dex (PO) 20 mg: first cycle
ASCT, autologous stem cell transplant; D, day; d/Dex, dexamethasone; HDT, high-dose therapy; Isa, isatuximab; IV, intravenous; NDMM, newly diagnosed multiple myeloma; PD, progressive disease; PO, oral; R/Len, lenalidomide; SC, subcutaneous; Te, transplant eligible; V/Bor, bortezomib; RVd is off label use in some countries according to the lenalidomide summary of product characteristics. 1. ClinicalTrials.gov: NCT03617731


Patients with MRD negativity at the end of induction therapy
OR 1.83 (95% CI 1.34–2.51)
Low number of not assessable/missing† MRD status: Isa-RVd (10.6%) and RVd (15.2%)
Isa-RVd is the first regimen to demonstrate a rapid and statistically significant benefit from treatment by reaching a MRD negativity of 50.1% at the end of induction and to show superiority vs. RVd in a Phase 3 trial Series1
GMMG
*P value derived from stratified conditional logistic regression analysis
†Missing NGF-MRD values were due to either patients’ loss to follow-up during induction therapy or to missing bone marrow samples or technical failures in measurement counted as non-responders, i.e. NGF-MRD positive CI, confidence interval; d, dexamethasone; Isa, isatuximab; ITT, intent-to-treat; MRD,

42 active sites; enrollment: Oct 7, 2020 ‒ Nov 15, 2021
Four 28-day cycles

Key eligibility criteria:
TE NDMM patients
aged <70 years
Stratification:
- Centralized FISH (standard risk/missing vs. high risk defined as del(17p) and/or t(4;14) and/or t(14;16); - ISS (I vs. II and III) R
4× KRd
K: 20 mg/m2 IV dd 1 cc 1 only; followed by 56 mg/m2 IV dd
8,15 cc 1 and dd 1,8,15 cc 2-4
R: 25 mg PO daily dd 1-21
d: 40 mg PO dd 1,8,15,22
4× Isa-KRd
Isa: 10 mg/kg IV dd 1,8,15,22 cc 1, followed by 10 mg/kg IV dd 1 and 15 cc 2 to 4.
K: 20 mg/m2 IV dd 1 cc 1 only; followed by 56 mg/m2 IV dd
8,15 cc 1 and dd 1,8,15 cc 2-4
R: 25 mg PO daily dd 1-21
d: 40 mg PO dd 1,8,15,22
MOBILIZATION
Cy: 2-3 g/m2 followed by G-CSF for stem-cell collection and MEL200-ASCT
MEL: 200 mg/m2 followed by ASCT
Post-ASCT consolidation
Four 28-day cycles
4× KRd
K: 56 mg/m2 IV dd 1,8,15
cc 5-8
R: 25 mg PO daily dd 1-21
d: 40 mg PO dd 1,8,15,22
Light consolidation
Twelve 28-day cycles
12× KRd
K: 56 mg/m2 IV dd 1,15
R: 10 mg PO dd 1-21
d: 20 mg PO dd 1,15
MRD by NGS
4× Isa-KRd
Isa: 10 mg/kg IV dd 1,15 cc 5-8
K: 56 mg/m2 IV dd 1,8,15
cc 5-8
R: 25 mg PO daily dd 1-21
d: 40 mg PO dd 1,8,15,22
MRD by NGS
12× Isa-KRd
Isa: 10 mg/kg IV d 1
K: 56 mg/m2 IV dd 1,15 R: 10 mg PO dd 1-21
d: 20 mg PO dd 1,15
MRD by NGS
MRD by NGS
PFS, progression-free
TE, transplant-eligible; NDMM, newly diagnosed multiple myeloma; FISH, fluorescence in situ hybridization; del, deletion; t, translocation; ISS, International Staging System stage; R, randomization; Isa, isatuximab; K, carfilzomib; R, lenalidomide; d, dexamethasone; IV, intravenous; dd, days; cc, cycles; PO, orally; Cy, cyclophosphamide; G-CSF, granulocyte colony-stimulating factor; MEL, melphalan; ASCT, autologous stem-cell transplantation; MRD, minimal residual disease; NGS, next-generation sequencing; survival.
OR 1.67, p=0.049
70% 80% 90% 100% 77% 67% P a ti e n t s ( % )
KRd (N=151) 0%
Isa-KRd (N=151)
High MRD compliance and sample quality (97-100% of sample evaluable at 10 -5 and 10-6 cut off.
Consistent MRD results were detected by next-generation flow
In the logistic regression analysis, ORs, 95% CIs, and p-values were adjusted for stratification factor. NGS, 10-5 NGS, 10-6
2.29, p<0.001
Isa-KRd (N=151)
KRd (N=151)
≥VGPR after consolidation was 94% in both arms; ≥CR 74% vs 72% and sCR 64% vs 67% in the IsaKRd vs KRd arms.


0%
NGS, 10-5 NGS, 10-6
Very high risk
0 HRCA 1 HRCA 2+ HRCA
2+ HRCA
1 HRCA was defined as the presence of one of the following high-risk cytogenetic abnormalities: del(17p13.1), t(4;14) (p16.3;q32.3), t(14;16) (q32.3;q23), gain(1q21), or amp(1q21); 2+ HRCA was defined as the presence of at least two high-risk cytogenetic abnormalities.
0 HRCA 1 HRCA
Very high risk


• Isa-KRd significantly increased post-consolidation 10-5 and 10-6 MRD negativity, as compared with KRd
• Isa-KRd significantly increased 10-5 and 10-6 MRD negativity after each treatment phase (Induction, Transplantation, Consolidation) .
• Isa-KRd consistently increased MRD negativity at 10-5 and 10-6 in all subgroups of patients, including high-risk and very high-risk disease.
• Isa-KRd treatment was tolerable, with a toxicity profile similar to that in previous reports.
• 10-6 MRD negativity cut-off is more informative.
• 1-year sustained MRD negativity will be available in 2024
• With a longer follow-up, this trial can offer the opportunity to explore the correlation between depth of MRD negativity and PFS/OS.
Dara-KRd
• Daratumumab 16 mg/m2 days 1,8,15,22 (days 1,15 C 3-6; day 1 C >6)
• Carfilzomib (20) 56 mg/m2 Days 1,8,15
• Lenalidomide 25 mg Days 1-21
• Dexamethasone 40mg PO Days 1,8,15,22 Dara-KRd x 4




MRD assessment by NGS


• We are transitioning to quadruplets in frontline eligible patients
• BUT the optimal length of a quadruplet is still to be determined!
• Transplant still has a role in MM even with long term use of novel agents
• Consolidation therapy may deepen responses and should be considered in patients who have not achieved VGPR
• MRD guided discontinuation may be possible in lower risk groups but not high risk patients

ASCO: What criteria are used to assess eligibility for autologous stem cell transplant (SCT)?
Recommendation
Patients should be referred to a transplant center to determine transplant eligibility
Evidence Rating
Type: Evidence based
Evidence quality: Intermediate, benefit outweighs harm
Strength of recommendation: Moderate
Chronologic age and renal function should not be the sole criteria used to determine eligibility for SCT.
Type: Evidence based
Evidence quality: Intermediate, benefit outweighs harm
Strength of recommendation: Moderate




Palumbo A Blood
IMWG score of 1 = Intermediate-Fit
3-year OS was 76% (HR=1.61; 95% CI 1.02-2.56; p=.042)
Toxicities 16.7% (HR 1.23, 95% CI 0.89-1.71; p=.217) and
Discontinuation 20.8% (HR=1.41; 95% CI 1.00-2.01; p=.052).
Engelhardt M. Haematologica 2016
Facon et al. Leukemia 2020
Bonanad et al. JGO 2015


‒ Patients were enrolled in MAIA from March 2015 through January 2017
D: 16 mg/kg IV
QW Cycles 1-2, Q2W Cycles 3-6, then Q4W thereafter until PD
Key eligibility criteria
• TIE NDMM
• ECOG PS
score 0-2
• CrCl ≥30 mL/min
R: 25 mg PO
Days 1-21 until PD
da: 40 mgb PO or IV
Days 1, 8, 15, 22 until PD
R: 25 mg PO Days 1-21 until PD
d: 40 mg PO
Days 1, 8, 15, 22 until PD
Cycles: 28 days
End-oftreatment visit (30 days after last dose)
Longterm follow-up
Primary endpoint
• PFS
Key secondary endpoints
• OS
• PFS2
• ORR
• CR/sCR rate
• MRD (NGS; 10–5)
MAIA is a multicentre, randomised, open-label, active-controlled, phase 3 study of D-Rd versus Rd alone in patients with NDMM who are transplant ineligible
aOn days when DARA


Median follow-up
Primary: 28.0 months1
Median follow-up
Update: 56.2 months
• D-Rd induced deeper responses with significantly higher rates of ≥CR and ≥VGPR, compared with Rd
• With >28 months of additional follow-up, responses deepened with continued DARA therapy
VGPR, very good partial response; PR, partial response. a
1. Facon T, et al. N Engl J Med 2019;380(22):2104-2115.

PFS rate
• D-Rd continued to demonstrate a significant PFS benefit, with median PFS not reached with DRd
• These data provide a new PFS benchmark in patients with NDMM who are transplant ineligible

Rd: median, NR 66.3% 53.1% 60-month OS rate
D-Rd: median, NR
D-Rd demonstrated a significant benefit in OS, with a 32% reduction in the risk of death, in patients with NDMM who are transplant ineligible
• Although ASCT remains the standard of care, use is likely to decline in patients who are 65-75 or with significant comorbidities
• Continuous therapy has resulted in better outcomes
• The balance of toxicity and efficacy is particularly important in this population
• ESPECIALLY with dexamethasone
• Most common approach is to select 2 agents from the 3 Novel Classes (PIs, IMiDs and MoAbs)
• Most will use DRD in standard risk patients
• Some may favor VRD in certain high risk patients
• DRD is more easily delivered and feasible
• D-VRD may well be a future standard of care even in these patients

Bortezomib
Lenalidomide
Carfilzomib
Panobinostat
Daratumumab Ixazomib
Now
VTD VRD KRD D-VMP DRD D-VRD SCT +/- More induction
Front line treatment
Induction Consolidation
Lenalidomide
Bortezomib Ixazomib
Lenalidomide + PI Carfilzomib
Combinations
Maintenance
Post consolidation
Pomalidomide Selinexor
Elotuzumab Isatuximab
Idecabtagene autoleucel
Ciltacabtagene autoleucel
Teclistamab
Talquetamab
Elranatamab
Rescue Relapsed
New
Isa-VRD
Isa-KRD D-KRD
CAR T or Bispecifics?
Daratumumab?
Novel CAR T Cell Therapies
Bispecific/Trispecific Antibodies
CelMod Agents
Venetoclax?
Modakafusp
Multiple small molecules

12:40 – 1:00 PM

1:00 – 1:10 PM
1:10 – 1:30 PM
March 2nd, 2024 – Agenda after lunch
Local Patient & Care Partner Panel, Sheri Baker, Patient & Jim Baker, Care Partner
Q&A
Maintenance Therapy, Andrew Portuguese, MD
1:30 – 1:40 PM Q&A
1:40 – 2:25 PM
Relapsed Therapies & Clinical Trials, Andrew Cowan, MD
2:25 – 2:35 PM Q&A
2:35 – 2:45 PM
2:45 – 3:00 PM
Closing Remarks
Coffee / Network








Valley MM Support Group Leaders
Sheri and Jim Baker Treasure
Andrew Portuguese, MD
Fred Hutchinson Cancer Center
Seattle, WA
• Discuss the principle of consolidation therapy and its application in myeloma
• Outline the major options for maintenance therapy
• Introduce the newer trend for the use of dual maintenance
• Provide an algorithm for maintenance based on risk status

• Induction: Intense and short term therapy with goal to achieve rapid remission
• Consolidation: Intense and shorter term therapy with goal of deep remission
• Maintenance: Less intense longer term therapy with goal of better PFS and OS
What does the Ideal Maintenance therapy look like?
• Deepen remission
• Prolong remission
• Easy to administer
• Minimal toxicity

CALGB 100104
(accrual 8/2005 – 11/2009)
INDUCTION
ASCT
1:1 RANDOMIZATION
“NO EVIDENCE OF PD”
Primary Endpoint: PFS
IFM 2005-02
(accrual 6/2006 – 8/2008)
INDUCTION
ASCT
1:1 RANDOMIZATION
“NO EVIDENCE OF PD”
Primary Endpoint: PFS
LEN: 2 COURSES
PLACEBO (n = 229)
LEN MNTCa (n = 231)
PLACEBO (n = 307)
LEN MNTCa (n = 307)
GIMEMA (RV-MM-PI-209) (accrual 11/2007 – 7/2009)
2 × 2 DESIGN
LEN + DEX × 4 INDUCTION
Primary Endpoint: PFS
LEN MNTCb (n = 67) NO TREATMENT (n = 68)
MPR: 6 COURSES
LEN MNTCb NO TREATMEN T ASCT
INTERIM ANALYSIS AND UNBLINDING
INTERIM AN Dec 2009 Jan 2010
CONTINUED
CROSSOVER BEFORE PD ALLOWED
CONTINUED TREATMENT
TREATMENT NO CROSSOVER BEFORE PD ALLOWED
ALL TREATMENT DISCONTINUED Jan 2011
PRIMARY ANALYSIS
CONTINUED TREATMENT
Target population of patients with NDMM who received LEN maintenance or placebo/no maintenance after ASCT
a Starting dose of 10 mg/day on days 1-28/28 was increased to 15 mg/day if tolerated and continued until PD.
b Patients received 10 mg/day on days 1-21/28 until PD.
CONTINUED TREATMENT
 ASCT, autologous stem cell transplant; LEN, lenalidomide; NDMM, newly diagnosed multiple myeloma; MNTC, maintenance; MPR, melphalan, prednisone, and Len; PD, progressive disease.
Attal et al ASCO 2016; McCarthy et al EHA 2016
ASCT, autologous stem cell transplant; LEN, lenalidomide; NDMM, newly diagnosed multiple myeloma; MNTC, maintenance; MPR, melphalan, prednisone, and Len; PD, progressive disease.
Attal et al ASCO 2016; McCarthy et al EHA 2016






3 randomized trials: 1,209 patients:
• Median follow up 6.6 years
• PFS 52.8 months for lenalidomide vs 23.5 in placebo
• PFS2 also prolonged 73.3 months vs 56.7 (ie not creating more aggressive clone)
• Median overall survival: 86 months v. not reached: P = 0.001
• Benefit for ≤ PR as well as VGPR/CR patients
• 29% discontinuation rate with lenalidomide
• Second primary malignancy rate higher at 6.1% vs 2.8% in placebo after PD McCarthy

Induction
NDMM
Treated on Myeloma XI induction protocols
N=1551 (TE=828; TNE=723)
Median follow-up: 27 months (IQR 13‒43)
Exclusion criteria
• Previous or concurrent active malignancies
Lenalidomide
10 mg/day, days 1‒21/28
Observation Maintenance
• Failure to respond to lenalidomide as induction IMiD, or development of PD
IQR, interquartile range; NDMM, newly diagnosed multiple myeloma; PD, progressive disease R 1:1

Significant improvement in PFS from 18 to 36 months, HR=0.45
Median PFS, months [95% CI]
Lenalidomide (n=857) 36 [31, 39]
Observation (n=694) 18 [16, 20]
HR=0.45; 95% CI 0.39, 0.52 Log-rank p<0.0001



474 NDMM patients, transplant-eligible and younger than 65 years
4x KCd
K: 36^ mg/m2 d 1-2,8-9,15-16
C: 300 mg/m2 d 1,8,15
d: 20 mg. d 1-2,8-9,15-16,2223
R1 1:1:1
4x KRd
K: 36^ mg/m2 d 1-2,8-9,15-16
R: 25 mg d 1-21
d: 20 mg. d 1-2,8-9,15-16,2223
Single ASCT
4x KCd
K: 36 mg/m2 d 1-2,8-9,15-16
C: 300 mg/m2 d 1,8,15
d: 20 mg. d 1-2,8-9,15-16,2223
Intensification with high-dose melphalan followed by autologous stem-cell reinfusion
4x KRd
K: 36 mg/m2 d 1-2,8-9,15-16
R: 25 mg d 1-21
d: 20 mg. d 1-2,8-9,15-16,2223
4x KRd
K: 36^ mg/m2 d 1-2,8-9,15-16
R: 25 mg d 1-21
d: 20 mg. d 1-2,8-9,15-16,2223
4x KRd
K: 36 mg/m2 d 1-2,8-9,15-16
R: 25 mg d 1-21
d: 20 mg. d 1-2,8-9,15-16,2223
4x KRd
K: 36 mg/m2 d 1-2,8-9,15-16
R: 25 mg d 1-21
d: 20 mg. d 1-2,8-9,15-16,2223
R2 1:1
R: 10 mg days 121, until progression or intolerance
K: 36 mg/m2 d 1, 2, 15, 16 up to 2 years*
R: 10 mg days 1-21, until progression or intolerance
^20 mg/m2 on days 1-2, cycle 1 only. *Carfilzomib 70 mg/m2 days 1, 15 every 28 days up to 2 years for patients that have started the maintenance treatment from 6 months before the approval of Amendment 5.0 onwards.
NDMM, newly diagnosed multiple myeloma, R1, first randomization (induction/consolidation treatment); R2, second randomization (maintenance treatment); ASCT, autologous stem-cell transplantation; K, carfilzomib; R, lenalidomide; C, cyclophosphamide; d, dexamethasone; KCd_ASCT, KCd induction-ASCT-KCd consolidation; KRd_ASCT, KRd induction-ASCT-KRd consolidation; KRd12, 12 cycles of KRd.
KRd_ASCT vs. KRd12 vs. KCd_ASCT
Median follow-up from Random 1: 51 months (IQR 46‒55)
Median follow-up from Random 2: 37 months (IQR 33‒42)
KRd_ASCT vs. KCd_ASCT: HR 0.54, 95% CI 0.38-0.78, p<0.001
KRd_ASCT vs. KRd12: HR 0.61, 95% CI 0.43-0.88, p=0.0084
Presented by Francesca Gay in 2021 ASCO Annual meeting
3-year PFS reported in the figure. Random 1, first randomization (induction/consolidation treatment); ASCT, autologous stem-cell trasplantation; K, carfilzomib; R, lenalidomide; C, cyclophosphamide; d, dexamethasone; KCd_ASCT, KCd induction-ASCT-KCd consolidation; KRd_ASCT, KRd induction-ASCT-KRd consolidation; KRd12, 12 cycles of KRd; Random 2, second randomization (maintenance treatment); p, p-value; HR, hazard ratio; CI, confidence interval.3-year progression-free survival
Median follow-up from Random 2: 37 months (IQR 33-42)
Presented by Francesca Gay in 2021 ASCO Annual meeting
Double
• It appears that dual maintenance therapy prolongs PFS
• This occurs in both standard risk AND high risk patients
• It further opens the door to other dual maintenance strategies currently being used and explored:
• Lenalidomide + Bortezomib
• Lenalidomide + Ixazomib
• Lenalidomide + Daratumumab
• Others??

• CASSIOPEIA randomized pts to no maintenance vs dara q 8 weeks
• Overall there was a benefit to having dara maintenance vs placebo
• However, if dara had been given at induction, that benefit did not seem to continue (ie If you had dara upfront, it didn’t add more to maintenance)
• However, PERSEUS added Dara to Lenalidomide for up to 2 years based on sustained MRD status
• There is still more to learn, but there is much more comfort use dara+len as maintenance, especially in higher risk patients

Induction Consolidation Maintenance
VRd
V: 1.3 mg/m2 SC
Key eligibility criteria
• Transplanteligible NDMM
• Age 18-70 years
• ECOG PS ≤2 1 : 1 r a n d o m i z a t i o n ( N = 7 0 9 ) a
Days 1, 4, 8, 11
R: 25 mg PO Days 1-21
d: 40 mg PO/IV
Days 1-4, 9-12
D-VRd
DARA: 1,800 mg
SCb
QW Cycles 1-2
Q2W Cycles 3-4
VRd administered as in the VRd group
4 cycles of 28 days
Primary endpoint: PFSc
T r a n s p l a n t
VRd V: 1.3 mg/m2 SC
Days 1, 4, 8, 11
R: 25 mg PO Days 1-21
d: 40 mg PO/IV
Days 1-4, 9-12
D-VRd
DARA: 1,800 mg
SCb
Q2W
VRd administered as in the VRd group
2 cycles of 28 days
Key secondary endpoints: Overall ≥CR rate,c overall MRD-negativity rate,d OS
D-R

R: 10 mg PO Days 1-28 until PD
DARA: 1,800 mg
SCb Q4W
R: 10 mg PO
Days 1-28
MRD positiv e MRD negativ e R
Continue D-R until PD
Discontinue DARA therapy only
28-day cycles
Discontinue DARA therapy only after ≥24 months of D-R maintenance for patients with ≥CR and 12 months of sustained MRD negativity
Abstracts Session, ASH 2023. Accessed from: https://ash.confex.com/ash/2023/webprogram/Paper191911.html
Restart DARA therapy upon confirmed loss of CR without PD or recurrence of MRD

Charlotte Pawlyn1,2, Tom Menzies3, Faith Davies4, Ruth de Tute5, Rowena Henderson3, Gordon Cook3,6 , Matthew Jenner7, John Jones8, Martin Kaiser1,2, Mark Drayson9, Roger Owen8, David Cairns3 , Gareth Morgan4, Graham Jackson10
On behalf of the Myeloma XI Trial Management Group and NCRI Haem-Onc Clinical Studies Group
 1) The Institute of Cancer Research, London, UK; 2) The Royal Marsden Hospital, London, UK; 3) Clinical Trials Research Unit, Leeds Institute of Clinical Trials Research, University of Leeds, Leeds, UK; 4) Perlmutter Cancer Center, NYU Langone Health, New York, US; 5) HMDS, Leeds Cancer Centre, Leeds Teaching Hospitals NHS Trust, Leeds, United Kingdom; 6) Leeds Cancer Centre, Leeds Teaching Hospitals NHS Trust, Leeds, UK; 7) University Hospital Southampton NHS Foundation Trust, Southampton, UK; 8) Kings College Hospital NHS Foundation Trust, London, UK; 9) Institute of Immunology and Immunotherapy, University of Birmingham, Birmingham, UK; 10) Department of Haematology, University of Newcastle, Newcastle-upon-Tyne, UK
1) The Institute of Cancer Research, London, UK; 2) The Royal Marsden Hospital, London, UK; 3) Clinical Trials Research Unit, Leeds Institute of Clinical Trials Research, University of Leeds, Leeds, UK; 4) Perlmutter Cancer Center, NYU Langone Health, New York, US; 5) HMDS, Leeds Cancer Centre, Leeds Teaching Hospitals NHS Trust, Leeds, United Kingdom; 6) Leeds Cancer Centre, Leeds Teaching Hospitals NHS Trust, Leeds, UK; 7) University Hospital Southampton NHS Foundation Trust, Southampton, UK; 8) Kings College Hospital NHS Foundation Trust, London, UK; 9) Institute of Immunology and Immunotherapy, University of Birmingham, Birmingham, UK; 10) Department of Haematology, University of Newcastle, Newcastle-upon-Tyne, UK
Randomisation
NDMM
Myeloma XI induction protocols and ASCT
N=1248
Median follow up: 44.7 months (IQR 32.4-62.7)
Maintenance R 1: 1
10mg/day, days 1-21/28
2 years 3 years
5 years 4 years

Observation
Median duration of lenalidomide therapy 28 cycles (range 1-96)
• Patients still on therapy 330/730 (45%)
• These data suggest an ongoing PFS benefit associated with continuing lenalidomide maintenance beyond at least 4-5 years in the overall patient population

• Even in patients with sustained MRD negativity, there is evidence of benefit from continuing lenalidomide maintenance for at least 3 years in total
• Randomised trials to address the impact of stopping lenalidomide maintenance in patients with sustained MRD negativity could be considered, at no earlier than 3 years
• In patients who are MRD +ve these data support continuing lenalidomide until disease progression
• No evidence of cumulative haematological toxicity was identified
• These findings emphasise the need for long term follow up of maintenance studies to enable the exploration of such questions
• There is a planned powered OS update of Myeloma XI in 2023
• Should post-transplant maintenance therapy be recommended for all patients?
• Yes
• Which agent should be used?
• Lenalidomide remains the standard of care – we may be adding daratumumab soon
• What is the optimal duration?
• Treatment until progression remains the standard of care
• What should patients with high-risk cytogenetics receive?
• Consider lenalidomide + proteasome inhibitor or daratumumab; clinical trial
• Should MRD status dictate maintenance therapy?
• Not outside of a clinical trial
• What about Second Primary Malignancies?
• They are real, require a discussion and monitoring, but are outweighed by benefit

Andrew Cowan, MD
Fred Hutchinson Cancer Center Seattle, WA

• Discuss an approach to treating relapsed myeloma based on patient, disease and treatment characteristics
• Review the important trend of using an aggressive approach in early treatment of myeloma
• Outline the key results from recent trials in early relapse
• Discuss the approach to late relapse and the use of novel therapies such as CAR T and bispecific antibodies

• It is not a simple algorithm of treatment #1 then 2 then 3…
• Leverage the benefit of multiple mechanisms of action in combination therapy
Categories:
• 1-3 prior lines
• Later Relapse
• Refractory to PI, IMiD and MoAb = Triple Class Refractory
Principles
1. Depth of Response matters…likely incorporate MRD soon
2. High risk vs standard risk…more aggressive Rx in high risk
3. Balance efficacy and toxicity…initially and constantly assess
4. Overcome drug resistance…change mechanism of action when possible
• Relapsed: recurrence (reappearance of disease) after a response to therapy
• Refractory: progression despite ongoing therapy
• Progression: change in M protein/light chain values
• Line of therapy: change in treatment due to either progression of disease or unmanageable side effects
• Note: initial (or induction) therapy + stem cell transplant + consolidation/ maintenance therapy = 1 line of therapy




• Nature of the relapse
– Biochemical vs symptomatic
• Risk stratification
– High-risk chromosomal abnormalities: del(17p), t(4;14), t(14;16)
• Disease burden
Therapy-Related
• Previous therapies
• Prior treatment-related adverse event
• Regimen-related toxicity
• Depth and duration of previous response
• Cost to patient
Patient-Related
• Renal insufficiency
• Hepatic impairment
• Comorbidities
• Preferences
• Social factors
– Support system
– Accessibility to treatment center
– Insurance coverage


RVD+ASCT+Lenalidomide Maintenance
Relapse 2
Relapse 1
Relapse 1 Relapse 2
Time: Years!
Relapse 3
Relapse 1

Not Refractory to Lenalidomide
Preferred options:
DRd (or KRd)
Alternatives:
DVD, Kd, DaraKd, IsaKd, IRd, Erd
When Dara, Isa, K not available: Rd, Vd, VTD, VCD, VMP
Refractory to Lenalidomide
Preferred options: PVd DaraKd
IsaKd
Second options
DaraVd;Kd
Other options:
KPd; DaraPd; Ipd
When Dara, isa, K or P not available: VCD, Vd, VMP
DKd,daratumumab/carfilzomib/dexamethasone; DPd, daratumumab/pomalidomide/dexamethasone; DRd, daratumumab/lenalidomide/ dexamethasone; DVd, daratumumab/bortezomib/dexamethasone; Elo–Rd, elotuzumab/lenalidomide/dexamethasone; Ipd, ixazomib/pomalidomide/dexamethasone; Ird, ixazomib/lenalidomide/dexamethasone; Isa–Kd, isatuximab/carfilzomib/dexamethasone; Kd, carfilzomib/dexamethasone; KPd, carfilzomib/pomalidomide/dexamethasone; KRd, carfilzomib/lenalidomide/ dexamethasone; PVd, pomalidomide/bortezomib/dexamethasone; Rd, lenalidomide/dexamethasone; SVd, selinexor/bortezomib/dexamethasone; VCd, bortezomib/cyclophosphamide/dexamethasone; Vd, bortezomib/dexamethasone; VMP, bortezomib/melphalan /prednisone; VTd, bortezomib/thalidomide/dexamethasone.
Moreau P, et al. IMWG Recommendations for RRMM. Lancet Oncol. 2021;22(3):e105-e118.
General Principles
Use mechanisms of action not previously used
Do not continue to use lenalidomide if progressing on len maintenance
Triplets are preferred over doublets
In real practice - most patients receiving VRD (Bortezomib-Lenalidomide-Dex) like regimens, 1st relapse is typically
Daratumumab + Pomalidomide + Dex (APOLLO)
Isatuximab + Pomalidomide + Dex (ICARIA)
Daratumumab + Carfilzomib + Dex (CANDOR)
Isatuximab + Carfilzomib + Dex. (IKEMA)
Selinexor + Bortezomib + Dex (BOSTON)

Not Refractory to Lenalidomide* Refractory to Lenalidomide*
Not refractory to CD38 moAB
Dara-refractory or Relapse while on CD38 moAB
Not refractory to CD38 moAB
Dara-refractory or Relapse while on CD38 moAB
DRd
KRd (preferred)
ERd, IRd (Alternatives)
DKd or Isa-Kd Or DPd or Isa-Pd
KCd or KPd (preferred)
VCd or EPd (Alternatives)
*Consider salvage ASCT in patients eligible for ASCT who have not had transplant before; Consider 2nd auto SCT if eligible and had >36 months response duration with maintenance to first ASCT
Drug Formulation Approval
Velcade (bortezomib)

Kyprolis (carfilzomib)

Ninlaro (ixazomib)
Revlimid (lenalidomide)*
Pomalyst (pomalidomide)*
XPOVIO (selinexor)




• IV infusion
• SC injection
• IV infusion
• Weekly dosing
• For relapsed/refractory myeloma
• For relapsed/refractory myeloma as a single agent, as a doublet with dexamethasone, and as a triplet with Revlimid or Darzalex plus dexamethasone
Once-weekly pill
Once-daily pill
• For relapsed/refractory myeloma as a triplet with Revlimid and dexamethasone
• For relapsed/refractory myeloma in combination with dexamethasone
Once-daily pill
• For relapsed/refractory myeloma in combination with dexamethasone
Once-weekly pill
• For relapsed/refractory myeloma as a triplet with Velcade and dexamethasone
*Black box warnings: embryo-fetal toxicity; hematologic toxicity (Revlimid); venous and arterial thromboembolism
IV, intravenous; SC, subcutaneous
• Velcade-Pomalystdex (VPd) vs Vd Regimens compared
• Kyprolis-Revlimiddex (KRd) vs Rd
Median progression-free survival favored
• VPd: 11 vs 7 months
• KRd: 26 vs 17 months
• Ninlaro-Rd (IRd) vs Rd
• XPOVIO-Velcadedex (XPO-Vd) vs Vd
Clinical considerations
• Consider for relapse on Revlimid
• VPd associated with more low blood counts, infections, and neuropathy than Pd
• KRd associated with more upper respiratory infections and high blood pressure than Rd
• IRd: 21 vs 15 months
• XPO-Vd: 14 vs 9 months
• IRd an oral regimen
• Gastrointestinal toxicities and rashes
• Lower incidence of peripheral neuropathy
• XPO-Vd associated with low platelet counts and fatigue with triplet, but less neuropathy than the Vd
Drug Formulation
Approval
Darzalex (daratumumab)

SC once a week for first 8 weeks, then every 2 weeks for 4 months, then monthly
• For relapsed/refractory myeloma as a single agent and as a triplet with Revlimid or Velcade or Kyprolis or Pomalyst plus dexamethasone
Empliciti (elotuzumab)

Sarclisa (isatuximab)

IV, intravenous; SC, subcutaneous
IV once a week for first 8 weeks, then every 2 weeks (or every 4 weeks with pom)
IV once a week for first 4 weeks, then every 2 weeks
• For relapsed/refractory myeloma as a triplet with Revlimid or Pomalyst and dexamethasone
• For relapsed/refractory myeloma as a triplet with Pomalyst or Kyprolis and dexamethasone
• Darzalex-Revlimiddex (DRd) vs Rd Regimens compared POLLUX
• Darzalex-Velcadedex (DVd) vs Vd
• Darzalex-Kyprolisdex (DKd) vs Kd
• Darzalex-Pomalystdex (DPd) vs Pd
Median progressionfree survival favored
• DRd: 45 vs 18 months
• DVd: 17 vs 7 months
• DKd: 29 vs 15 months
• DPd: 12 vs 7 months
Clinical considerations
• Consider for relapses from Revlimid or Velcade maintenance
• DRd associated with more upper respiratory infections, low blood white blood cell counts, and diarrhea
• Consider for patients who are Revlimid-refractory without significant neuropathy
• DVd associated with more low blood cell counts
• Consider for younger, fit patients who are doublerefractory to Revlimid and Velcade
• DKd associated with more respiratory infections
• Sever side effects (possibly fatal) in intermediate fit patients 65 and older
• Consider in patients who are double-refractory to Revlimid and a proteasome inhibitor (Velcade, Kyprolis, Ninlaro)
• Severe low white blood cell counts
• Empliciti-Revlimiddex vs Rd Regimens compared
• EmplicitiPomalyst-dex vs Pd
Median progressionfree survival favored
• Empliciti-Rd: 19 vs 15 months
• Empliciti-Pd: 10 vs 5 mos
• Sarclisa-Pomalyst-dex vs Pd
Clinical considerations
• Consider for nonRevlimid refractory, frailer patients
• Overall survival benefit with Empliciti-Rd
• Empliciti-Rd associated with more infections
• Consider for patients refractory to Revlimid and a proteasome inhibitor (Velcade, Kyprolis, Ninlaro)
• Sarclisa-Pd: 12 vs 7 mos
• Sarclisa-Kyprolis-dex vs Kd
• Sarclisa-Kd: 41 vs 19 mos
• Consider for patients refractory to Revlimid and a proteasome inhibitor (Velcade, Kyprolis, Ninlaro)
• Sarclisa-Pd associated with severe low white blood cell counts, more dose reductions, upper respiratory infections, and diarrhea
• Consider for patients refractory to Revlimid and Velcade
• Sarclisa-Kd associated with higher MRD negativity rates
• Sarclisa-Kd associated with severe respiratory infections
Refractory to IMiD, PI, Anti-CD38
Combinations with Cyclophosphamide that do not have IMiD, PI, Anti CD38 (e.g., KCd)
Anti BCMA strategy
Anti-BCMA
Bispecific
BCMA CAR-Ts
Refractory to IMiD, PI, Anti-CD38, Alkylators, and Anti-BCMA
Existing drugs:
Elotuzumab
Selinexor
Venetoclax
Bendamustine
VDT PACE
New Drugs: Iberdomide, Mezigdomide
New bispecifics (Cevostamab, Talquetamab)
New CAR-Ts
New Monoclonals
New ADCs

Class Drug Formulation Approval
Nuclear export inhibitor XPOVIO (selinexor)
Twice-weekly pill
Antibody-drug conjugate
Chimeric antigen receptor (CAR) T cell
Blenrep (belantama b mafodotin)*
Abecma (idecabtage ne vicleucel)
Bispecific antibody
2.5 mg/kg IV over approximately 30 minutes once every 3 weeks
300 to 460 × 106 genetically modified autologous CAR T cells in one or more infusion bags
• For relapsed/refractory myeloma in combination with dexamethasone (after at least 4 prior therapies and whose disease is refractory to at least 2 PIs, at least 2 IMiDs, and an anti-CD38 mAb
• For relapsed/refractory myeloma (after at least 4 prior therapies including an antiCD38 mAb, a PI, and an IMiD
• For relapsed/refractory myeloma (after 4 or more prior lines of therapy, including an IMiD, a PI, and an anti-CD38 mAb
Tecvayli (Teclistamab) Step up dosing then weekly SQ For relapsed/refractory myeloma (after 4 or more prior
CAR T cell Carvykti (ciltacabtag ene autoleucel)
0.5 to 1.0 × 106
genetically modified autologous CAR T cells/kg of body weight
Talquetamab lines of therapy, (PI, an IMiD, and an anti-CD38 mAb Elranatamab
• For relapsed/refractory myeloma (after 4 or more prior lines of therapy, including a PI, an IMiD, and an anti-CD38 mAb
IMiD, immunomodulatory agent; PI, proteasome inhibitor; mAb, monoclonal antibodyBispecific antibodies
• Cevostamab, Alnuctamab, ABBV383, and others
• Target BCMA, GPRC5D, or FcRH5 on myeloma cells and CD3 on T cells
• Redirects T cells to myeloma cells
Cereblon E3 ligase modulators (CELMoDs)
• Iberdomide
• Targets cereblon
• Enhances tumoricidal and immune-stimulatory effects compared with immunomodulatory agents
Small molecule inhibitors
• Venetoclax
• Targets Bcl-2
• Induces multiple myeloma cell apoptosis
Collect patient’s white blood cells
Isolate and activate T cells Engineer T cells with CAR gene
Tumor cell

Targeting element (eg, CD19, BCMA, CD20)
Spacer
Expand CAR T cells
Infuse same patient with CAR T cells
Activity
CD19
Viral vector with CAR DNA CARengineere d T cell
Transmembrane domain
Costimulatory domain (eg, CD28 or 4-1BB)
CD3 ( �� essential signaling domain)
Median manufacturing time: 17-28 days
Patients undergo lymphodepleting (and possibly salvage/bridging) therapy
Majors. EHA 2018. Abstr PS1156. Lim. Cell. 2017;168:724. Sadelain. Nat Rev Cancer. 2003;3:35.
Brentjens. Nat Med. 2003;9:279. Park. ASH 2015. Abstr 682. Axicabtagene ciloleucel PI. Tisagenlecleucel PI. Slide
1 Apheresis

2 (Manufacturing) Patients return home

3 Lymphodepletion (chemotherapy)

1 day 4–6 weeks
Immune cells from the patient are collected
4 Infusion

3 days
Standard of care therapy is permitted until CAR T cells are ready for infusion
5

2 weeks
Fludarabine and Cytoxan are used to create “immunologic space” to CAR T cells to expand
27-month PFS
ORR, overall response rate; PR, partial response; VGPR, very good partial response; CR, complete response; sCR, stringent complete response; MRD, minimal residual disease; PFS, progression-free survival
KarMMa Trial. Munshi NC et al. N Engl J Med. 2021;384:705.
CARTITUDE-1 Trial. Berdeja JG et al. Lancet. 2021;398:314; Martin T et al. J Clin Oncol. June 4, 2022 [Epub ahead of print].

Cytokine release syndrome (CRS)


Neurotoxicity (ICANS)

Onset
Duration
Symptoms
19 days after CAR T-cell infusion
29 days after CAR T-cell infusion
511 days 317 days
• Fever
• Difficulty breathing
• Dizziness
• Nausea
• Headache
• Rapid heartbeat
• Low blood pressure
• Headache
• Confusion
• Language disturbance
• Seizures
• Delirium
• Cerebral edema
Management
• Actemra (tocilizumab)
• Corticosteroids
• Supportive care
• Antiseizure medications
• Corticosteroids
*Based on the ASTCT consensus; †Based on vasopressor; ‡For adults and children >12 years;
§For children ≤12 years; ‖Only when concurrent with CRS
Xiao X et al. J Exp Clin Cancer Res. 2021;40(1):367. Lee DW et al. Biol Blood Marrow Transplant. 2019;25:625; Shah N et al. J Immunother Cancer. 2020;8:e000734.

Median (range)
MM CAR-T infusion volume in 2021 10-50 (<5,50-100)
Number of FDA approved CAR-T slots given per month 1 (0-4)
Patients on wait list (FDA approved CAR-T) 20 (5-100)
Duration a patient is on waiting list 6 (2-8) months
Outcomes of patients on wait list
(0-50%)
(0-50%) 25% (0%-75%)
FDA approved CAR-T CAR-T trial non-CAR-T trial hospice or death 25% (0%-64%)
Survey of 20 centers. Responses from 17 centers.
CAR T-cell therapy
Autologous stem cell transplantation
Patient’s cells collected Yes Yes
Types of cells collected T cells* Stem cells†
Collected cells are genetically engineered in a lab Yes No
Patient given chemotherapy before cells are infused back into patient Yes, lymphodepleting therapy Yes, melphalan
When in the course of myeloma is this usually done?
Side effects of treatment
After multiple relapses As part of initial treatment
Cytokine release syndrome; confusion Fatigue, nausea, diarrhea
*An immune cell that is the “business end” of the system, in charge of maintaining order and removing cells.
†Precursor cells that give rise to many types of blood cells. We actually collect CD34+ve cells.

There are currently 3 approved bispecific antibodies:
Teclistamab (Tecvayli)
Talquetamab (Talvey)
Elranatamab (Elrexfio)
Bispecific antibodies are also referred to as dual specific antibodies, bifunctional antibodies, or T-cell engaging antibodies
Bispecific antibodies can target two cell surface molecules at the same time (one on the myeloma cell and one on a T cell)
Many different bispecific antibodies are in clinical development; none are approved for use in myeloma
Availability is off-the-shelf, allowing for immediate treatment




BCMA, GPRC5D, or FcRH5
Examples:
• Elranatamab
• Teclistamab
• TNB-303B (ABBV-383)
• REGN5458
• Cevostamab
• Talquetamab
Drug Formulation Approval
Tecvayli (teclistama b)*

Step-up dosing† the first week then once weekly thereafter by subcutaneous injection
• For relapsed/ refractory myeloma (after 4 or more prior lines of therapy, including an IMiD, a PI, and an anti-CD38 mAb)
IMiD, immunomodulatory agent; PI, proteasome inhibitor; mAb, monoclonal antibody
*Black box warning: cytokine release syndrome; neurologic toxicities
†Patients are hospitalized for 48 hours after administration of all step-up doses.
Tecvayli is available only through a restricted distribution program.
Median duration of response 18.4 months
CR or better median DOR not reached (95% CI: 16.2–NE)
• Overall median DOR of 18.4 months (95% CI: 14.9–NE), and was not yet mature with data from 71 patients (68.3%) censored
Patients at risk Overall
CR or better
Overall median DOR
18.4 months (95% CI: 14.9–NE)
• 12-month event-free rate:
• Overall:
• Patients with CR or better:
68.5% (95% CI: 57.7–77.1)
80.1% (95% CI: 67.6–88.2)
Teclistamab was well tolerated; discontinuations and dose reductions were infrequent
• 2 patients (1.2%) discontinued due to AEs (grade 3 adenoviral pneumonia; grade 4 PML)
• 1 patient had dose reduction at cycle 21
• The most common AEs were CRS and cytopenias
• Infections occurred in 126 (76.4%) patients (grade 3/4: 44.8%)
• 123 patients (74.5%) had evidence of hypogammaglobulinemia a
• There were 19 deaths due to AEs, including 12 COVID-19 deaths
• 5 deaths due to teclistamab-related AEs:
• COVID-19 (n=2)
• Pneumonia (n=1)
• Hepatic failure (n=1)
• PML (n=1)

T P

• Median duration of followup was 12.0 mos (range 0.3–32.3)
• ORR = 64% (95% CI, 50–75); CR/sCR rate = 38% (21/55)
• 54% (7/13) of patients with prior BCMA-directed therapy achieved response
• For responders (n = 35), median TTR = 36 days (range 7–262)
Data cutoff 9/30/2022
Swimmer plot depicts disease assessments relevant to first response, confirmation of response, deepening of response, and best response. Mutational analysis was filtered on functional mutations annotated in OncoKB and normal allele frequency <5% in paired peripheral blood mononuclear cell samples. * Prior anti-BCMA ADC. * Prior BCMA-targeted CAR-T. MR = minimal response; NE = not evaluable; PD = progressive disease; Q2W = every 2 weeks; REL = relapse; SD = stable disease; TTR = time to response.
Raje

*Calculated from n =106 responders in each group.
QW = weekly.
Chari
ORR was similar for QW and Q2W schedules
• Triple-class refractory: 72.6% and 71.0%
• Penta-drug refractory: 71.4% and 70.6%
• ORR was consistent across subgroups including baseline ISS stage III disease, baseline cytogenetic risk, number of prior therapies, and belantamab exposure, except among patients with baseline plasmacytomas
100%
80%
60%
40%

CRS
79.0 % 72.4 %
Grade 2: 21 (14.7%)
Grade 3: 3 (2.1%)
Grade 2: 25 (17.2%) P
20%
0%
Grade 189 (62.2%)
Grade 3: 1 (0.7%)
Grade 179 (54.5%)
405 μg/kg SC QW 800 μg/kg SC Q2W (n = 143) (n = 145)
*AEs were graded by CTCAE v4.03 with 2–3 step-up doses.
Chari A, et al.
Blood. 2022;140 (suppl 1): 384-387.
CRS events
• Most CRS events were grade 1/2 and largely confined to step-up doses and first full dose
Non-hematologic AEs
• Low rates of grade 3/4 nonhematologic AEs
• Low discontinuation rates due to AEs
Most common AEs were CRS, skinrelated events, nail-related events, and dysgeusia
Hematologic AEs
• Most high-grade AEs were cytopenias
• Cytopenias were generally limited to first few cycles
*Based on a recent sampling
• Cytokine release syndrome (CRS)
• Neurotoxicity (ICANS)
• Usually occurs within first 1–2 weeks
• Frequency (all grade and grade 3–5) higher with CAR T
• Cytopenias
• Target unique
• For example, rash, taste disturbance seen with GPRC5D, but not with BCMA
• Infections
• Incidence for bispecifics at RP2D not yet known
• Viruses: CMV, EBV
• PCP/PJP
• Ongoing discussions regarding prophylactic measures
IVIG
Anti-infectives
CAR T-cell therapy Bispecific antibody
Approved product Abecma, Carvykti Tecvayli
Efficacy ++++ +++
How given
Where given
Notable adverse events
One-and-done
Academic medical centers
CRS and neurotoxicity
IV or SC, weekly to every 3 weeks until progression
Academic medical centers
CRS and neurotoxicity
Cytokine release syndrome +++ ++
Neurotoxicity ++ +
Availability Wait time for manufacturing
Off-the-shelf, close monitoring for CRS and neurotoxicity
CAR T and bispecific antibodies are very active even in heavily pretreated patients.
Side effects of CAR T cells and bispecific antibodies include cytokine release syndrome, confusion, and low blood counts, all of which are treatable.
Abecma and Carvykti are only the first-generation CAR T cells and target the same protein. Different CAR Ts and different targets are on the way. Bispecific antibodies represent an “off-the-shelf” immunotherapy; Tecvayli was approved in October 2022, and now Talvey and Elrexfio in August 2023
Several additional bispecific antibodies are under clinical evaluation.
Bortezomib
Lenalidomide
Carfilzomib
Panobinostat
Daratumumab Ixazomib
Now
VTD VRD KRD D-VMP DRD D-VRD SCT +/- More induction
Front line treatment
Induction Consolidation
Lenalidomide
Bortezomib Ixazomib
Lenalidomide + PI Carfilzomib
Combinations
Maintenance
Post consolidation
Pomalidomide Selinexor
Elotuzumab Isatuximab
Idecabtagene autoleucel
Ciltacabtagene autoleucel
Teclistamab
Talquetamab
Elranatamab
Rescue Relapsed
New
Isa-VRD
Isa-KRD D-KRD
CAR T or Bispecifics?
Daratumumab?
Novel CAR T Cell Therapies
Bispecific/Trispecific Antibodies
CelMod Agents
Venetoclax?
Modakafusp
Multiple small molecules

• Provide the rationale for clinical trials
• Outline the phases of clinical trials
• Discuss the risks and benefits of clinical trials
• Preview important clinical trials in myeloma
Remember some of the important principles of clinical trials:
• The drive of research has brought us to where we are
• No one is expected to be a “guinea pig” with no potential benefit to them
• Research is under very tight supervision and standards
• Open, clear communication between the physician and the patient is fundamental
• Every patient is unique and must be viewed that way
• Benefits of trials are numerous and include:
• Early access to “new” therapy
• Delay use of standard therapy
• Contribution to myeloma world – present and future
• Financial access to certain agents
• Must be balanced with potential risks
• “toxicity” of side effects
• Possibility of lack of efficacy
• Clinical trials translate results of basic scientific research into better ways to prevent, diagnose, or treat cancer
• The more people that take part, the faster we can:
• Answer critical research questions
• Find better treatments and ways to prevent cancer



Identify a target for therapy in the laboratory
Confirm the anticancer activity in laboratory and animal studies
Clinical trials (human studies) to determine safety, dosing and effectiveness
The whole process costs millions of dollars and years of effort!
• Most agents are tested in lab models
• Various “myeloma cell lines” = in vitro
• Next step is animal model
• We are more like mice than you think!!
• Earliest study in phase I is called “First in Human”
• Often uses extremely low dose of drug to ensure safety


Tests safety Tests how well treatment works Compares new treatment to standard treatment
• All patients receive the experimental therapy
• Phase 1 trials find the optimal dose of a new drug or drug combination
• Patients get higher doses as the study continues
• Determine side effects of new drugs or combinations
• Explore how the drug is metabolized by the body
• Important for all stages of myeloma
• Determine if a new drug or combination is effective against the cancer
• May be added to a phase 1 study once the ideal dose is found
• Patients usually receive the experimental therapy
• In some cases, the study may include two “arms” comparing either two different doses or a different treatment (another combination of drugs)
• Highest form of clinical evidence. Typically a large number of patients are required…usually required for full FDA approval
• Patients receive either an experimental therapy (one or more drugs) or the current standard treatment
• The patient is randomly assigned to a treatment—a process called randomization
• Neither the physician or the patient can determine which treatment is given
• May be placebo controlled, if no standard treatments are available
• Very closely monitored for effectiveness and side effects
•
Each cancer clinical trial has a written detailed study design called a protocol that includes:
• Why the clinical trial is needed
• Purpose of the clinical trial
• What drug or drug(s) are being tested, with a treatment and follow-up schedule
• Safety measures throughout the clinical trial program
• How outcomes will be measured
• Who is eligible for the clinical trial
• How the clinical trial will be organized, one site or multiple sites
• If the clinical trial is a multi-site trial, all participating physicians must follow the same protocol
Possible benefits:
• Patients will receive, at a minimum, the best standard treatment
• If the new treatment or intervention is proven to work, patients may be among the first to benefit
• Patients have a chance to help others and improve cancer care
Possible risks:
• New treatments or interventions under study are not always better than, or even as good as, standard care
• Even if a new treatment has benefits, it may not work for every patient
• Health insurance and managed care providers do not always cover clinical trials
• Be unaware of clinical trials
• Lack access to trials
• Fear, distrust, or be suspicious of research
• Have practical or personal obstacles
• Face insurance or cost problems
• Be unwilling to go against their physicians’ wishes
• Not have physicians who offer them trials
• Have a disconnect with their healthcare team
• There has been a lack of diverse representation in clinical trials in myeloma. In the U.S., approximately 20% of all myeloma patients are of African descent, but only 5%–8% of patients in myeloma clinical trials are of African descent.
• This is significant for the following reasons:
All patients of all races and ethnicities should be able to benefit from clinical trials.
Diverse patient representation in clinical trials is required to ensure that the outcomes are applicable to all patients.
• Reasons for underrepresentation in clinical trials are complex and include systemic racism, accessibility of clinical trials, sensitivity to diversity by medical professionals, misconduct in medicine in the past, the lack of trust in the system, and more.
Doctors might:
• Lack awareness of appropriate clinical trials
• Be unwilling to “lose control” of a person’s care
• Believe that standard therapy is best
• Be concerned that clinical trials add administrative burdens
How does the study work? How often will I need to see my doctor or visit the cancer center?
Will I need to undergo additional tests?
What is currently known about the new drug or combination?


What benefits can I expect?
What side effects should I expect? Who should I notify if I have side effects?


Can I take my vitamins or other medications?
Can I get the treatment with my local doctor?
Will my insurance pay for my participation in the clinical trial?




• Discuss whether or not you are eligible for a clinical trial with your physician
• Work with your physician to determine the best trial for you
• Meet with the clinical research nurse or trials coordinator to discuss the trial
• Carefully review the provided “Informed Consent”
• Describes the study and any potential safety concerns related to the experimental medication


• SO many areas being studied right now including…
• CAR T Cell therapy (used earlier in myeloma, new targets, faster manufacturing, even allo CAR T!)
• Novel Bispecific Therapies (with new targets) and even Trispecifics
• New molecules – CelMods, …
And MANY more!








Patient and Family Seminars
San Francisco – April 12th & 13th, 2024 – Crowne
Plaza Foster City – San Mateo

Los Angeles – August 16th & 17th, 2024 – Hilton Los
Angeles/Universal City
Regional Community Workshops
May 18th, 2024 – Salt Lake City RCW; please check
myeloma.org for more information on registration

Thank you for attending today’s program!
March 23rd, 2024,
International Myeloma Foundation’s Regional Community WorkshopSeattle